- Home
- Blog
- Detoxification
- Endometriosis and functional medicine: Evidence‑informed insights for clinical practice
Endometriosis and functional medicine: Evidence‑informed insights for clinical practice
0%
Endometriosis affects approximately 10–15% of women of reproductive age and remains one of the most challenging chronic inflammatory conditions encountered in women’s health practice. Characterised by the presence of endometrial-like tissue outside the uterine cavity, it is an oestrogen-dependent, immune-modulated and inflammatory disorder associated with chronic pelvic pain, infertility and significant quality-of-life burden.
Although conventional management, including hormonal suppression, NSAIDs and surgical excision remains central to care, recurrence rates and persistent symptoms highlight the need for a broader systems-based approach.
This article explores the underlying mechanisms driving endometriosis and outlines evidence-informed nutritional and functional medicine strategies to support clinical outcomes.
Understanding inflammatory, hormonal and environmental drivers to support patient care
The traditional explanation of retrograde menstruation alone does not account for why only some women develop endometriosis. Current evidence supports a multifactorial model involving immune dysfunction, inflammatory signalling, altered hormone metabolism and environmental influences.
- Immune dysregulation
While retrograde menstruation is common, impaired immune surveillance appears central to implantation and survival of ectopic endometrial tissue. Alterations in macrophage activity, NK cell function and inflammatory cytokine signalling have been reported. Some authors have questioned whether endometriosis may exhibit autoimmune-like features (Gleicher et al., 1987).
The peritoneal environment plays a significant role in lesion establishment and inflammatory amplification (Young et al., 2013).
- Oestrogen dependence and progesterone resistance
Endometriosis is strongly oestrogen-sensitive. Lesions demonstrate local oestrogen production via increased aromatase expression and altered progesterone receptor signalling (Attar & Bulun, 2006; Patel et al., 2014).
Imbalances may be driven by:
- Increased peripheral aromatisation
- Impaired hepatic detoxification
- Enhanced enterohepatic recirculation
- Environmental xenoestrogen exposure
Shifts in oestrogen metabolism toward proliferative pathways may contribute to lesion growth.
- Chronic inflammatory signaling
Pro-inflammatory mediators particularly prostaglandin E2 (PGE2) contribute to pain, angiogenesis and further aromatase activation (Burney & Giudice, 2012).
Arachidonic acid metabolism via COX-2 is central to inflammatory amplification. This pathway is also the primary pharmacological target of NSAIDs, which, while effective for short-term pain relief, carry gastrointestinal and cardiovascular risks with prolonged use.
- Environmental toxicant exposure
Endocrine-disrupting chemicals (EDCs) including dioxins and polychlorinated biphenyls (PCBs) have been investigated for potential roles in disease initiation and progression (Rier & Foster, 2002; Guo, 2004; Anger & Foster, 2008).
Although causality in humans remains debated, mechanistic evidence supports their ability to alter immune signalling, cytokine production and hormone receptor expression.
A functional clinical framework
An integrative approach focuses on modulating inflammation, supporting detoxification pathways, restoring gut balance and optimising micronutrient status.
-
Supporting hepaticoestrogendetoxification
The liver metabolises oestrogens primarily via Phase I and Phase II pathways (glucuronidation and sulfation), rendering them water-soluble for excretion.
However, beta-glucuronidase-producing gut bacteria may deconjugate oestrogens, facilitating enterohepatic recirculation and potentially contributing to oestrogen dominance.
Clinical strategies
- Increase dietary fibre to promote elimination
- Address constipation
- Support beneficial microbiota
- Consider calcium D-glucarate (reduces beta-glucuronidase activity)
- Encourage cruciferous vegetables (source of indole compounds including diindolylmethane [DIM])
DIM has been shown to favourably influence oestrogen metabolism toward protective 2-hydroxylated metabolites (Zeligs & Bradlow, 2006; Dalessandri et al., 2004).
Supportive nutrients may include:
- Magnesium
- Vitamin B6 (as P5P)
- B-complex vitamins
- Amino acids for conjugation
- Phosphatidylcholine
- Silymarin (milk thistle extract)
- Anti-inflammatory dietary intervention
Dietary modification remains one of the most impactful clinical tools.
Key principles
Reduce:
- Refined sugars
- Ultra-processed foods
- Excess omega-6 vegetable oils
- Red meat (source of arachidonic acid)
- Excess alcohol
Increase:
- Brassica vegetables
- High-fibre plant foods
- Oily fish
- Ground flaxseed
- Filtered water
- Essential fatty acid balance
Western diets often display omega-6:omega-3 ratios of 10–25:1, whereas physiological balance may be closer to 1–4:1.
Excess omega-6 fatty acids increase PGE2 production, reinforcing inflammatory signalling. Omega-3 fatty acids (EPA/DHA) demonstrate anti-inflammatory effects and have been shown to reduce IL-8 secretion in women with and without endometriosis.
Sources include:
- Oily fish
- Flaxseed
- Krill oil (phospholipid-bound form for enhanced bioavailability)
- Nutrientoptimisation& immune support
Nutrient deficiencies may exacerbate inflammatory and hormonal dysregulation.
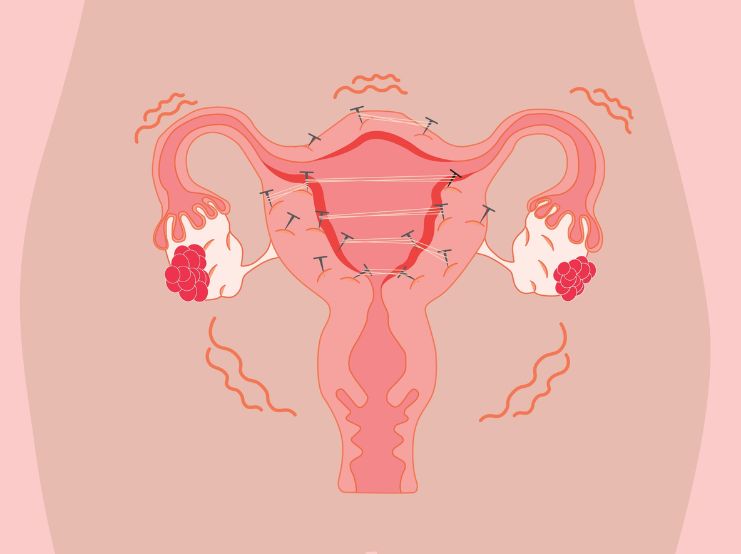

Key nutrients in clinical practice
Magnesium
Acts as an enzymatic cofactor, supports smooth muscle relaxation and modulates nociceptive signalling.
Vitamin E
May inhibit arachidonic acid pathways and reduce lesion-associated pain.
Vitamin C and bioflavonoids
Support immune regulation and antioxidant defense.
Beta-carotene
Enhances immune function
- Botanical support for inflammatory pain
Selective modulation of inflammatory pathways may offer advantages over non-selective COX inhibition.
Alpha acids extracted from Humulus lupulus (hop) have demonstrated selective COX-2 modulation and reduction of pro-inflammatory mediators in preliminary research, without affecting COX-1 homeostatic functions.
Emerging clinical data suggest potential benefits for inflammatory pain, though larger controlled trials are warranted before routine integration into standard protocols.
- Environmental medicine considerations
Encourage reduction of endocrine disruptor exposure:
- Avoid heating food in plastic containers
- Limit canned foods (BPA exposure)
- Choose glass or stainless steel storage
- Reduce pesticide burden where feasible
- Consider low-tox personal care products
While the evidence base continues to evolve, reducing cumulative endocrine burden aligns with broader hormone-balancing strategies.
Clinical integration: A practical checklist
- Assess inflammatory load and fatty acid balance
- Support hepatic detoxification and gut integrity
- Optimise micronutrient status (magnesium, B6, antioxidants)
- Address bowel regularity
- Reduce environmental endocrine disruptor exposure
- Provide collaborative care alongside gynaecology
Conclusion
Endometriosis is a systemic inflammatory, immune-modulated and hormone-dependent condition requiring a multi-layered clinical strategy.
By addressing upstream drivers: inflammation, oestrogen metabolism, gut dysbiosis, nutrient insufficiency and environmental burden, practitioners can provide meaningful adjunctive support alongside conventional care.
References
Anger DL, Foster WG. The link between environmental toxicant exposure and endometriosis. Front Biosci. 2008;13:1573–1582.
Attar E, Bulun SE. Aromatase inhibitors: the next generation of therapeutics for endometriosis? Fertil Steril. 2006;85(5):1307–1318.
Burney RO, Giudice LC. Pathogenesis and pathophysiology of endometriosis. Fertil Steril. 2012;98(3):511–519.
Dalessandri KM, Firestone GL, Fitch MD, et al. Effect of 3,3′-diindolylmethane on urinary hormone metabolites in postmenopausal women. Nutr Cancer. 2004;50(2):161–167.
Gleicher N, el-Roeiy A, Confino E, Friberg J. Is endometriosis an autoimmune disease? Obstet Gynecol. 1987;70(1):115–122.
Guo SW. The link between exposure to dioxin and endometriosis: a critical reappraisal. Gynecol Obstet Invest. 2004;57(3):157–173.
Patel B, Elguero S, Thakore S, et al. Role of nuclear progesterone receptor isoforms in uterine pathophysiology. Hum Reprod Update. 2014;21(2):155–173.
Rier S, Foster WG. Environmental dioxins and endometriosis. Toxicol Sci. 2002;70(2):161–170.
Young VJ, Brown JK, Saunders PTK, Horne AW. The role of the peritoneum in the pathogenesis of endometriosis. Hum Reprod Update. 2013;19(5):558–569.
Zeligs MA, Bradlow HL. Diindolylmethane (DIM) and its role in oestrogen metabolism. Proc Am Assoc Cancer Res. 2006;47.
Karen Devine, CNHC Registered Nutritionist and Naturopath, is a Colonic Hydrotherapist and Functional Medicine practitioner (AFMCP). Since 2007, she has provided technical support for Nutri-Link Ltd, advising practitioners and patients on nutritional products. Trained at The Plaskett College, Karen has run her own clinic since 1999 and continues to consult privately. She regularly attends postgraduate seminars to stay current in functional medicine. Karen has appeared on The Spa of Embarrassing Illnesses...
- Allergies (3)
- Lifestyle Medicine (35)
- Menopause Support (0)
- Minerals (2)
- Nervous System (7)
- Phytonutrients (2)
- Stress Management (16)
- Thyroid (2)
- Vitamins (30)
- Women's Health (3)
- Men's Health (0)
- Fertility & Pregnancy (2)
- Coronavirus (23)
- Diets (74)
- Vitamin C (3)
- Zinc (3)
- Vitamin D (10)
- Sleep (8)
- Fatigue (3)
- Vegan (1)
- Vitamin B12 (1)
- Product Updates (2)
- Respiratory (0)
- Bone Health (3)
- Immune Health (69)
- Joint Health (2)
- Musculoskeletal (0)
- Essential Fatty Acids (9)
- Metabolism (15)
- Mental Health (31)
- Digestive Health (34)
- Probiotics (13)
- Weight management (13)
- Ageing (8)
- Food (31)
- Brain Health (28)
- Adrenal Health (3)
- Antioxidants (1)
- Cardiovascular Health (11)
- Children's Health (1)
- Detoxification (6)
- Energy (3)
- Eye Health (0)